| ☰ |
How to edit bonds
Changing bond type
If single bond type is selected in the toolbar, clicking on an existing bond will increase the bond type from single to double to triple and back to single bond. Clicking on a stereo bond or dashed bond will change it to a single bond.
If double bond type is selected in the toolbar, clicking on an existing bond will change it to a plain double bond. Clicking on an existing double bond will change the appearence of the double bond from plain to upper bar and lower bar double bond.
If stereo bond type is selected in the toolbar, clicking on an existing bond will change it to stereo bond. Clicking on a stereo bond will change it's direction.
If special bond type is selected, clicking on an existing bond will change it to the corresponding special bond. More special bonds can be selected in the popover by clicking on the disclosure button
- If the delocalized bond tool is selected as special bond, clicking on an existing delocalized bond changes its appearance successively (delocalization shown above, below or invisible).
- If the dative (coordinative) bond tool is selected as special bond, clicking on a bond of this type will change the direction of the arrow tip.
Changing bond attributes like type, line-width, color
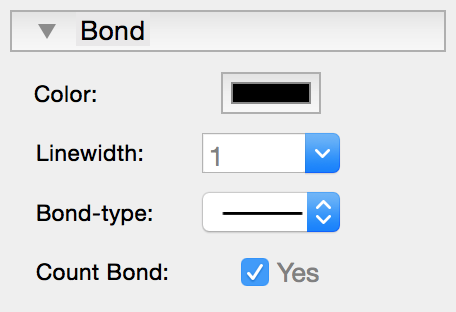 |
By right-clicking on a bond or by right-clicking on selected bonds a popover is shown where you can change the attributes of the selected bonds. If the option "Count bond(s)" is set to "NO", the selected bonds will not be taken into account for the calculation of free valences or charge. |
Justifying bonds
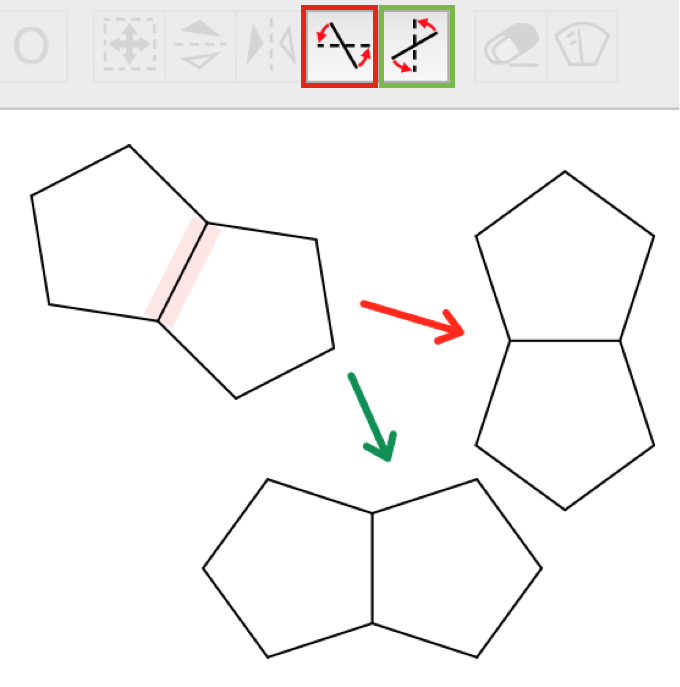 |
Molecules and Groups can be rotated horizontally or vertically in respect to a bond. Choose the correspondend tool in the toolbar and click on a bond. |
Bond types
 single bond
single bond
 double bond centered
double bond centered
 double bond above
double bond above
 double bond below
double bond below
 delocalized bond above
delocalized bond above
 delocalized bond below
delocalized bond below
 delocalized bond with invisible delocalization
delocalized bond with invisible delocalization
 triple bond
triple bond
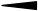 right sided stereo bond above
right sided stereo bond above
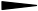 left sided stereo bond above
left sided stereo bond above
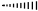 right sided stereo bond below
right sided stereo bond below
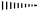 left sided stereo bond below
left sided stereo bond below
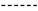 single dashed bond
single dashed bond
 wavy bond
wavy bond
 right sided dative (coordinative) bond
right sided dative (coordinative) bond
 left sided dative (coordinative) bond
left sided dative (coordinative) bond